How to Verify FDA Registration for Chinese Dental Suppliers
Published On:
For medical device distributors, pharmacy chains, and Amazon FBA sellers, sourcing oral care products from overseas is a highly profitable strategy. However, the dental supply chain is heavily regulated. Importing effervescent denture cleansers, retainer tablets, or dental adhesives requires strict adherence to United States customs and health regulations.
When searching for an FDA registered dental supplier China, many buyers fall into the trap of accepting forged documents or relying on unverified trading companies. A single compliance failure can result in seized shipments, suspended Amazon ASINs, and severe legal liabilities.
Verifying Dental Manufacturers
Demand the Owner/Operator Number: Legitimate manufacturers will instantly provide their 7-digit FDA Owner/Operator Number or Establishment Registration Number for public verification.
Search the Official FDA Database: Always cross-reference the provided number directly on the U.S. FDA’s “Establishment Registration & Device Listing” official website to ensure the factory is actively registered for the current year.
Identify Trading Companies vs. Direct Factories: Ensure the factory name on the FDA registration perfectly matches the name on the commercial invoice and the ISO 13485 certificate to avoid unauthorized middlemen.

The Financial Risks of Unverified Dental Manufacturers
The U.S. Food and Drug Administration (FDA) treats products like denture cleansers and dental adhesives as medical devices (typically Class I or Class II). When a shipping container arrives at a U.S. port, Customs and Border Protection (CBP) electronically reviews the manufacturer’s FDA registration status.
If you partner with a supplier holding a fake, expired, or mismatched FDA registration, your business faces immediate financial damage:
Customs Seizures: CBP will issue a hold, and your inventory will be detained or destroyed at the port at your expense.
Amazon FBA Suspensions: Amazon’s product safety team routinely audits health and personal care listings. Failing to provide a valid FDA establishment number and Safety Data Sheet (SDS) will lead to immediate listing suspension and stranded inventory.
Supply Chain Disruption: Replacing a rejected batch takes a minimum of 60 to 90 days, causing massive stockouts and lost market share.
Step-by-Step Guide: How to Verify an FDA Registered Dental Supplier in China
Do not accept a PDF certificate at face value. In the manufacturing sector, PDFs are easily edited. You must verify the data at the source.
Step 1: Request the Exact Registration Numbers
Ask your prospective supplier for two specific pieces of information:
Their Owner/Operator Number (usually a 7-digit number).
Their Establishment Registration Number (FEI number, often assigned after the initial registration process is finalized).
If the supplier hesitates, claims the information is “confidential,” or says they will “provide it later,” terminate the negotiation. Real factories use their FDA registration as their primary marketing tool and will provide it immediately.
Step 2: Access the Official FDA Database
Navigate to the official U.S. FDA website. Locate the “Establishment Registration & Device Listing” database. This is a public, free-to-use search engine maintained by the U.S. government.
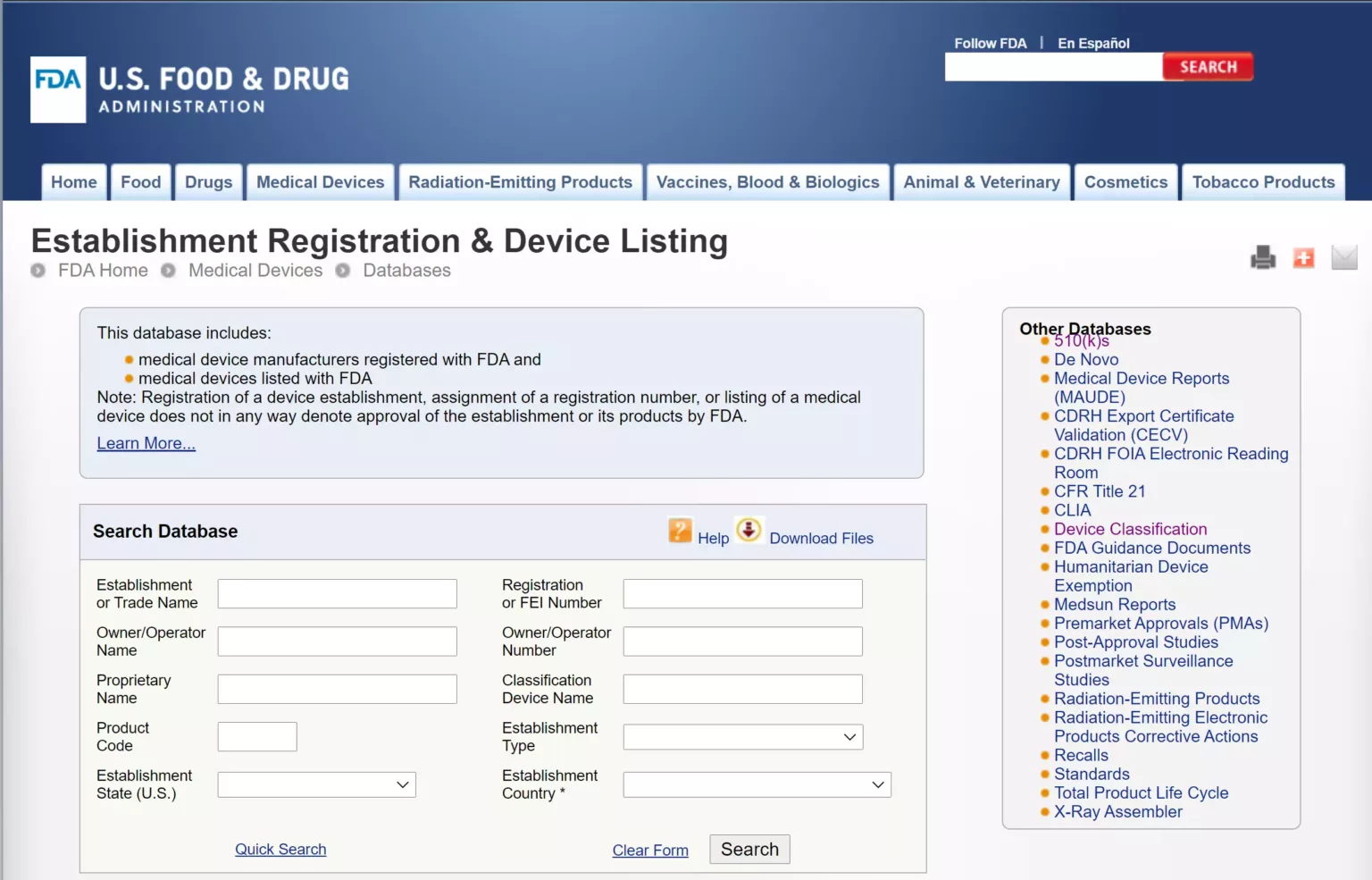
source: FDA Establishment Registration & Device Listing search portal
Step 3: Cross-Reference the Data
Input the supplier’s Owner/Operator Number into the search bar. When the results generate, verify the following data points:
Company Name: Does the registered name match the company you are paying?
Facility Address: Does the registered address match the physical factory location in China?
Status: Is the registration active for the current calendar year? (FDA registrations must be renewed annually between October 1 and December 31).
Device Listing: Click on the company profile and look at the products they are authorized to manufacture. If you are buying denture cleaning tablets, the device name “cleanser, denture” or a similar precise classification must be listed.
FDA “Registered” vs. FDA “Approved”: Understanding the Trap
A common red flag in B2B sourcing is a supplier claiming their denture tablets are “FDA Approved.”
In the United States regulatory framework, the FDA approves new pharmaceutical drugs and high-risk Class III medical devices (like pacemakers). For Class I and most Class II items—such as denture toothbrushes, cleaning tablets, and retainer cases—the FDA only requires the facility to be registered and the device to be listed.
If a Chinese supplier aggressively markets their dental accessories as “FDA Approved” and shows you a certificate with an FDA logo, they are likely a trading company that does not understand international medical regulations. The FDA explicitly prohibits the use of its logo on private company certificates or packaging.
Manufacturer vs. Trading Company: A Critical Verification Step
Many buyers mistakenly verify an FDA number, only to realize later that the number belongs to a source factory, while the company they are paying is an unverified middleman (trading company). This breaks the chain of custody and invalidates your compliance.
Use this table to evaluate your supplier’s structural integrity:
| Verification Metric | Direct OEM Manufacturer (e.g., ITS Dental Care) | Middleman / Trading Company |
|---|---|---|
| FDA Registration Name | Matches the commercial invoice perfectly. | Different from the invoice name (using another factory's ID). |
| ISO 13485 Certification | Holds their own medical QMS certificate for manufacturing. | Cannot provide a manufacturing ISO certificate in their name. |
| Facility Audit | Welcomes video tours of their automated pressing and packaging lines. | Refuses live video tours or only shows a standard office. |
| Formulation Control | Can customize chemical formulas, colors, and blister sizes in-house. | Offers limited, fixed options and cannot alter technical specs. |
| Unit Cost | Factory-direct pricing, protecting your retail margins. | Inflated pricing with an added broker markup. |
Why ITS Dental Care is the Safest Choice for B2B Procurement
Finding a reliable FDA registered dental supplier China should not be a gamble. Since 2007, ITS Dental Care has operated as a true source manufacturer specializing in senior oral care and denture maintenance products. We understand that your brand’s reputation and financial stability depend on strict supply chain compliance.
We do not just meet the baseline requirements; we engineer our operations around global medical standards. When you partner with us, you secure a supply chain built on:
FDA Registration: We maintain active, accurate facility registration and device listings, ensuring your cargo clears U.S. customs without friction.
ISO 13485 Certification: Our facility operates a strictly audited Quality Management System specifically designed for medical devices.
CE Compliance: We provide full legal access for buyers expanding into the European Economic Area.
Sedex Certification: We guarantee ethical labor practices and supply chain transparency, a mandatory requirement for modern retail chains.
100,000-Class Cleanrooms: Our effervescent denture tablets and zinc-free adhesive creams are manufactured in heavily controlled environments, guaranteeing consistent water activity levels and zero contamination.
When Amazon or customs authorities request your compliance paperwork, we provide standard Safety Data Sheets (SDS), Certificates of Analysis (COA), and direct proof of our factory registrations to protect your ASINs and your inventory.
Frequently Asked Questions (FAQ)
Q1: What happens if my supplier’s FDA registration expires while my goods are on a cargo ship?
A: If the registration lapses before the shipment clears U.S. customs, the cargo will be detained. It is critical to work with a mature factory that systematically renews its FDA facility registration every Q4. At ITS Dental Care, our compliance team handles all annual renewals proactively.
Q2: Can a Chinese dental supplier use the same FDA number for multiple different products?
A: The facility uses one Owner/Operator Number. However, every distinct product category (e.g., denture cleansers vs. plastic denture bath cases) must have its own separate Device Listing under that facility’s profile. Always verify that the specific product you are buying is listed.
Q3: Is an FDA registration enough to start selling on Amazon FBA?
A: No. While FDA registration is required for customs clearance, Amazon will also demand a professional Safety Data Sheet (SDS) and sometimes an exemption sheet for effervescent or chemical products. As an experienced OEM, we supply these technical documents upfront with your order.
Q4: How can I verify that you are the real factory and not a trading company?
A: We encourage all prospective B2B buyers to request our ISO 13485 and FDA documents, which clearly state our company name and exact physical address. We also welcome digital facility tours of our automated production lines.
Secure Your Dental Supply Chain Today
Do not risk your capital and brand reputation on unverified suppliers. Build your private label oral care business on a foundation of proven medical compliance and factory-direct efficiency.
We invite procurement managers and brand owners to verify our credentials and explore our manufacturing capabilities by visiting our About Us page.
If you are ready to review our product quality firsthand, browse our full catalog on our Products page. Contact the ITS Dental Care OEM team today to request our FDA registration details, a direct factory quote, and your free product samples.
Private Label Denture Cleansing Tablets
This is the absolute best product to start with. Denture cleansing tablets are highly effective, easy to use, and require zero technical support. Buyers drop a tablet into water, and it cleans their dentures in 3 minutes or overnight. From an FBA perspective, a box of 120 tablets is small, lightweight, and easy to pack into standard shipping boxes. You can easily create a private label brand and offer different variations, such as “Mint Flavor,” “Smoker Stain Remover,” or “Fast Dissolving.”
Zinc-Free Denture Adhesive Cream
Many older denture adhesives use zinc, which has been linked to health risks if overused. In 2026, consumers are actively searching for “Zinc-Free” and “Safe” alternatives. Launching a medical-grade, zinc-free denture adhesive cream provides a strong, all-day hold for seniors while addressing their health concerns. The small tube size makes it another winner for keeping Amazon fulfillment fees low.
Retainer and Clear Aligner Cleansers
Do not ignore the younger market. Millions of teenagers and adults use clear aligners and nighttime retainers. They cannot use standard toothpaste, as it scratches the plastic. Selling specialized “Retainer Cleaning Tablets” targets a younger, high-spending demographic. You can design modern, lifestyle-focused packaging that appeals directly to this audience.

Denture Bath Cases (The Perfect Bundle Item)
Selling a physical plastic case on its own might have lower margins, but it is the perfect item for a “Bundle Strategy.” By packaging a high-quality, leak-proof denture bath case together with a 30-day supply of your cleaning tablets, you create a unique ASIN. Bundling helps you avoid direct price wars with competitors and increases your average order value (AOV).
Get in Touch with ITS Dental
Have questions or need more information? Contact us today, and our dedicated team will assist you promptly.


ITS Dental is a trusted manufacturer of high-quality denture care products, including denture cleaning tablets, adhesive creams, and retainer cleaning solutions, offering customized services for global partners.